Responsible research assessment topics to consider before you apply
The Responsible research assessment highlights areas important to the HRB agenda, such as Open Access, Public and Patient Involvement in research (PPI), Gender equality, How we monitor and evaluate, as well as other relevant policies and practices. They should inform how you frame an application.
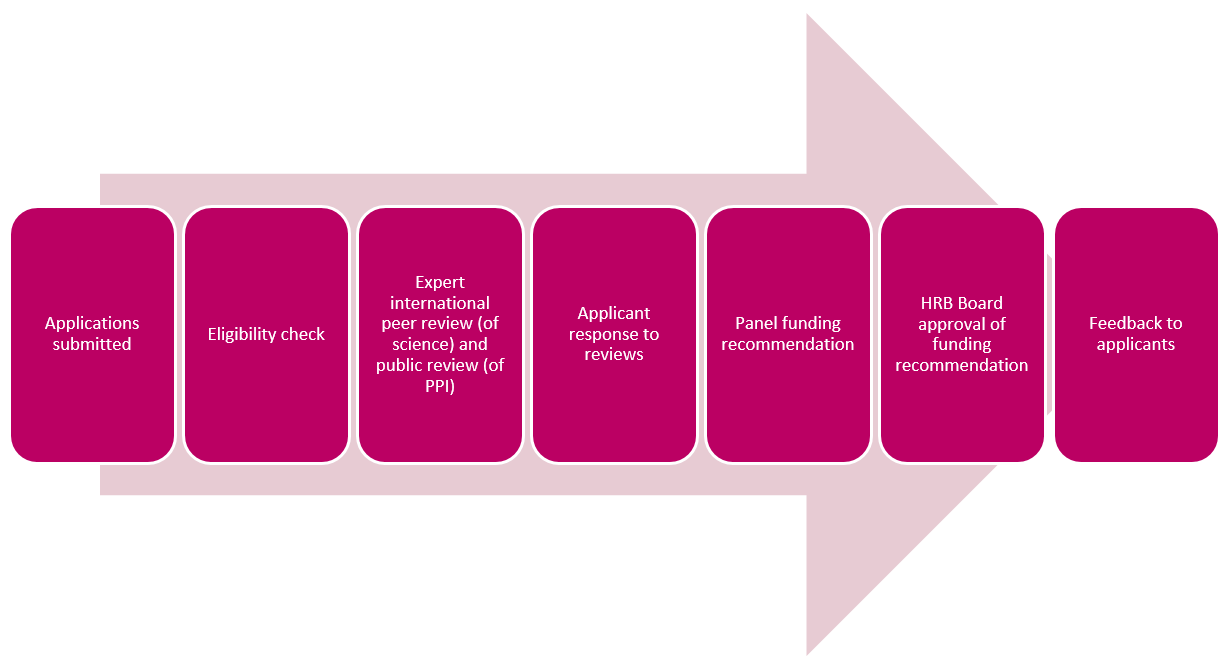
Tips for writing a grant application
Writing a grant application takes time and planning. Here we share some tips on how to write a good grant application, such as what to consider before you start writing, what reviewers look for in an application and the importance of writing for reviewers who don’t know the Irish system. We also look at the right to respond option and some of the reasons why grant applications are rejected.
Tips for writing a grant application
Useful links
We have put together some external resources you may find helpful in the preparation of an application. You will find useful links on areas such as study design for interventions, study registration, reporting, clinical research infrastructures, PPI, bio banking, data management, animals in research and gender issues amongst others.
View Resources